Investigator grants make up a huge portion of clinical trial costs, yet mid-study, most sponsor teams have difficulty answering the question: What do I actually owe my sites right now?
In a recent webinar, we walked through exactly why that gap exists—and what it takes to close it.
The Real Problem Is a Data Problem
The data needed to get a full picture of your investigator grants lives in three different places: Site Clinical Trial Agreements (CTAs), EDC systems, and site payments reports provided by CROs. Contacted prices for work performed is included in CTAs. A record of what clinical trial activity has been performed is in the EDC systems, and what payments have been invoiced are in payments reports. The problem, not only do these systems of record not talk to one another, but they use different naming conventions, report with different timing, and have different levels of granularity. What one system calls "Visit 3 – Screening" another calls "SV3" and a third records as a line-item invoice with no visit reference at all. There is no VLOOKUP or INDEX MATCH that can stitch these data sets together at scale.
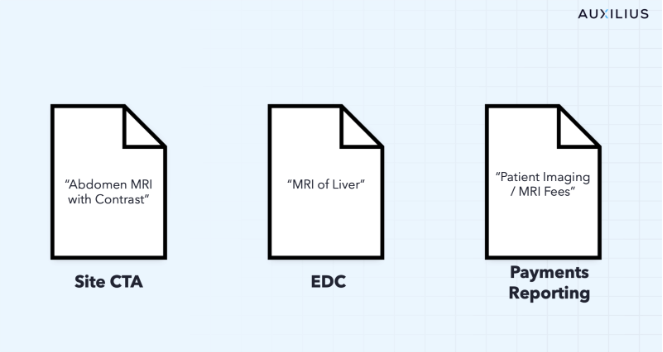
What Gets Missed When Payments Reports Drive the Picture
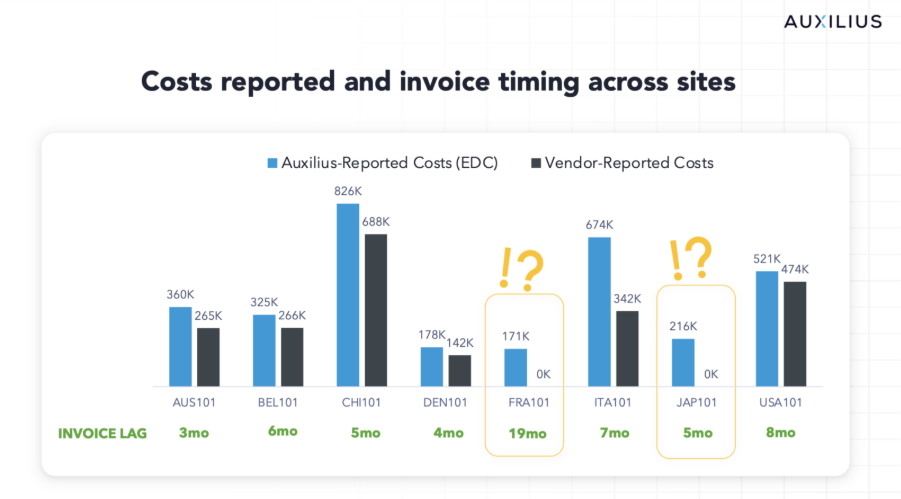
The most significant finding from our case study: a 44% average variance between real-time clinical data-based costs from Auxilius and what vendors were reporting (based on site invoices).
The culprit is invoice lag. For example, French sites in this case study showed $171,000 in incurred expense in Auxilius without anything invoiced to-date. Why? These sites typically invoice on average 19 months after work performed.
A Structured Model Changes What's Possible
Auxilius’s integrated platform creates a common taxonomy across CTAs, EDC, and payments data—applying contracted costs accounting for amendment timing, patient groups, treatment arms, and cohorts. The result is that you can build your own longitudinal, transaction-level view of investigator spend: life-to-date, by site, visit type, invoiceable category, and region.
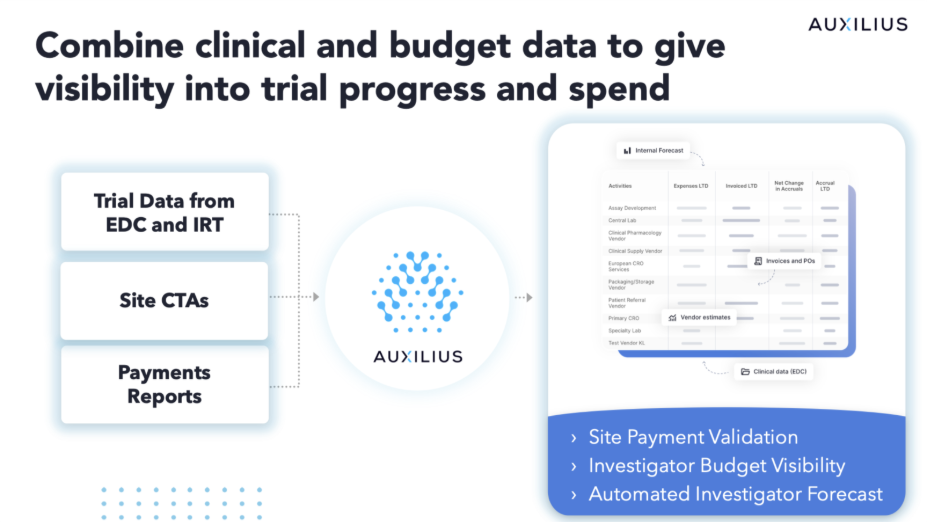
That data model allows sponsors to have a much more current view of their investigator grants, months ahead of lagging vendor reporting. This allows sponsors to better understand not only how much they owe sites, but also ensure sites are being paid correctly and on time.
Why This Matters Beyond Reconciliation
Investigator grant visibility isn't just an operational concern, it's a financial one. Finance teams are managing escrows, upfront payments, and end-of-study true-ups, often against CRO forecasts built on averages that don't update with enrollment or site mix. Understanding where patients are enrolling, and whether those sites are high- or low-cost, directly affects budget confidence, cash runway, and capital planning.
Getting this right also protects site relationships – especially for small or newer sites. Accurate, on-time payment matters most to sites with limited billing infrastructure, the ones least equipped to handle sponsor billing processes. If sponsors can identify and support sites that might be behind on invoicing (and getting paid), there’s a much better chance they can build a site relationship that lasts for the long term.
A reconciled, model-driven view of investigator spend isn't a nice-to-have. In complex global studies, it's the only way to know what you've actually committed, what's been incurred, and what's still coming. If your accruals are built on payments data alone, there's a meaningful chance you have an incomplete picture of your study financials.
Want to see how this applies to your portfolio? Get in touch.
.png)